
Need More Information?
If you are interested in prodisc and would like to learn more about the technology from a Centinel Spine representative, please fill in the form below.
ABOUT prodisc
prodisc is a Total Disc Replacement (TDR) technology platform that offers a surgical treatment proven to maintain spinal balance and motion, decelerate adjacent level reoperations, and accelerate the return to normal activities.
It is the only total disc replacement system in the U.S. approved for two-level use in the lumbar spine
Centinel Spine is currently the only company in the United States to offer Total Disc Replacement devices for both the cervical and lumbar spine:
*Not available in the U.S. at this time

Not all products are available in all markets.
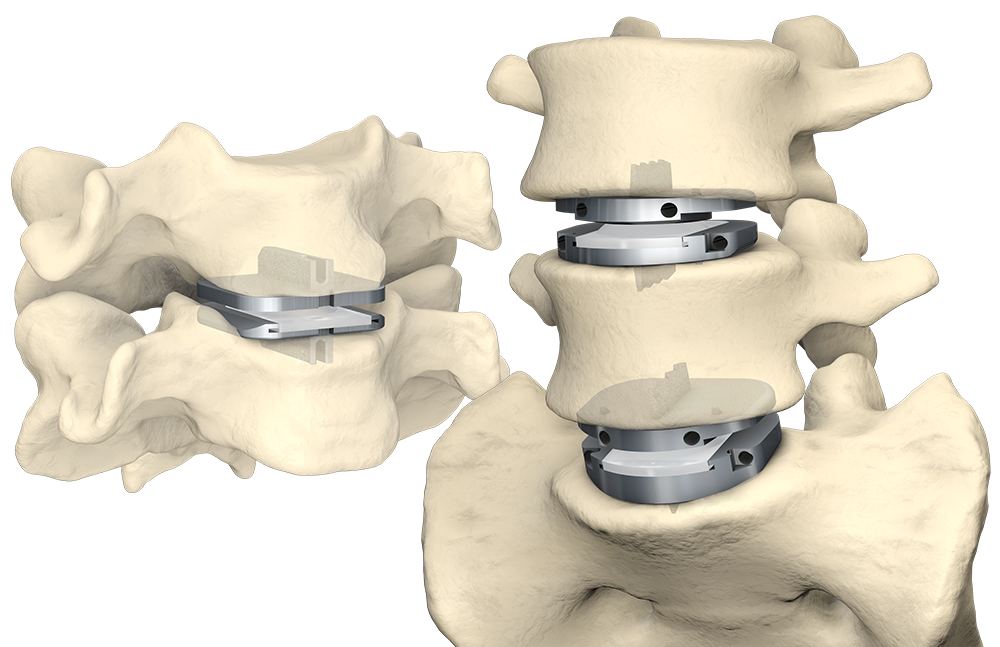
prodisc DESIGN
The prodisc semi-constrained design and fixed center of rotation (CoR) provides controlled and predictable segmental motion1 and limits hypermobility.2
Predictable Motion
The anatomy/physiology of the facet joint requires rotation about the CoR of the motion segment for proper movement of the joint structures. If a TDR implant can translate in the Anterior-Posterior plane in the absence of flexion/extension, then the facet joint may be loaded in shear.
The prodisc ball and socket has been designed to maintain a fixed center of rotation. This provides for flexion/extension of the motion segment, while at the same time, stabilizing and protecting the segment from further degeneration and shear1.
The constrained and controlled segmental motion of the prodisc design also limits hypermobility and allows the coupled motion of translation with flexion/extension—sparing the facets from distraction or impingement.2

prodisc TRAINING
If you are interested in training or hands-on courses for the prodisc technology platform, please visit the Centinel Spine med+ educational portal.
Centinel Spine's med+ program is dedicated to educating surgeons on the advantages of addressing spinal disease through anterior access of the spine with Centinel Spine's broad array of technologies—including the prodisc® Total Disc Replacement and STALIF® Integrated Interbody™ fusion devices.
prodisc USAGE
The 1st implantation of a prodisc L took place in 1990, and the 2nd generation design received US PMA approval in 2006.
With such a lengthy clinical history and global usage, the prodisc line of total disc replacements are the most widely studied TDRs in the world.
30+
30+ Year Clinical History with Worldwide Usage
prodisc design has been validated with almost 225,000 device implantations worldwide3 and more than 540 published papers4
2L
The only total disc replacement system in the U.S. approved for two-level use in the lumbar spine
Almost 225,000 device implantations and a reported reoperation rate of less than 1%
prodisc STUDIES
No other disc replacement system has been studied more than prodisc.
With more than 540 published papers, reporting on experiences with over 13,000 patients, the clinical data for prodisc strongly supports its safety and effectiveness. Here are just a few key takeaways from these numerous clinical studies:
After 7 Years with prodisc C vs. Patients that Received a Fusion (ACDF)5
After 5 Years with prodisc L vs. Patients that Received a Fusion (ALIF)6
Shown in Biomechanical Study of prodisc C vs. a Plate-Based Fusion (ACDF)7
1 Motion Analysis of a Mobile-Core Cervical Disc Prosthesis, RM. Havey, S. Khayatzadeh, L. Voronov, G. Carandang, FM. Phillips, AG. Patwardhan. Presented at ISASS 2018, April 11, Toronto, Canada.
2 prodisc Ball and Socket Design History, The PRODISC Book: Total Disc Replacement in the Lumbar spine, R. Bertagnoli, MD, T. Marnay, MD. Copyright 2003.
3 Data on file at Centinel Spine compiled from Spine Solutions, Synthes Spine, DePuy Synthes, and Centinel Spine.
4 Search performed on Pubmed, Embase, Ovid Medline® covering 1988 – 2018.
5 Janssen ME, et al, ProDisc-C Total Disc Replacement Versus ACDF for Single-Level Symptomatic Cervical Disc Disease, JBJS, 2015, 97:1738-47.
6 Zigler, J, Delamarter R, Five-year results of the prospective, randomized, multicenter, Food and Drug Administration investigational device exemption study of the prodisc L total disc replacement versus circumferential arthrodesis for the treatment of single-level degenerative disc disease, J Neurosurg Spine, 2012, 17:493-501.
7 Crawford NR, et al, Biomechanics of a Fixed-Center of Rotation Cervical Intervertebral Disc Prosthesis, Int J Spine Surg. 2012; 6: 34–42.
Watch this short primer on Centinel Spine and its unique and extraordinary place as a catalyst of change in the spine industry—with pioneering technologies and a clinical history that have led to successes ranging from PGA champions to a growing list of surgeon-patients.
 SEE MORE VIDEOS
SEE MORE VIDEOS