
Not Available for Sale in All Markets


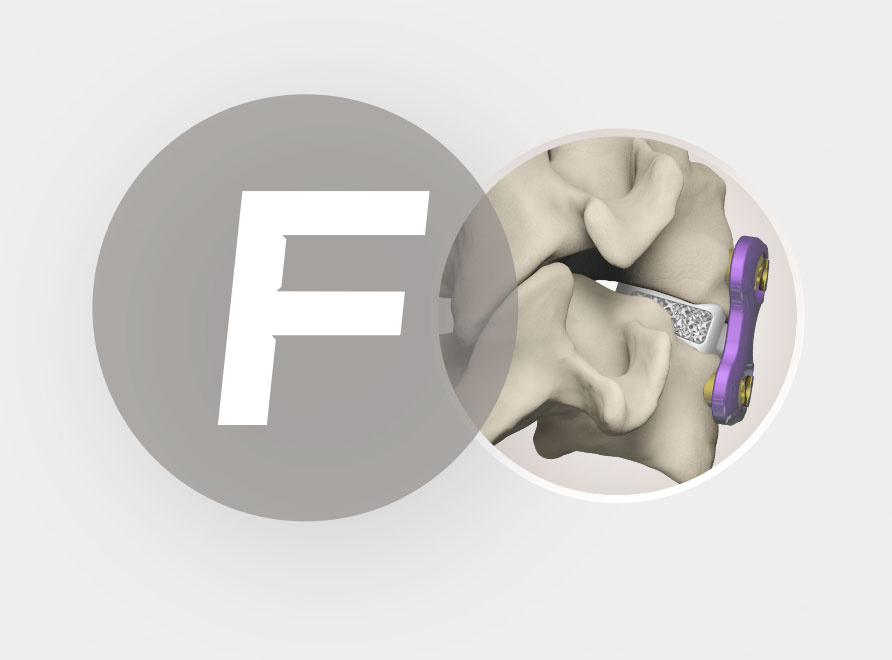



| Widths | A/P Depths | Anterior Heights | Posterior Heights |
| 14mm-18mm | 12mm-16mm | 4.5mm-11.5mm | 3mm-10mm |

Watch this three minute primer on Centinel Spine and its unique and extraordinary place as a catalyst of change in the spine industry—with pioneering technologies and a clinical history that have led to successes ranging from PGA champions to a growing list of surgeon-patients.
 SEE MORE VIDEOS
SEE MORE VIDEOS